-
Modern motorcycles cannot run without a battery.
-
Their technology has advanced much over the years.
-
Finding out about how they work is just as important as choosing one and maintenance.
There was a point in time when a motorcycle didn’t require a battery to start, relying instead on points and condenser ignition system, but now batteries are getting ever more advanced in order provide power to starter, lighting system, electronic ignition (ECU) and coils, riding aids, other electrical and electronic systems and accessories.
It’s the battery that’s powering all those systems at the start and when the motorcycle’s charging system can’t provide enough power for example when the bike is idling for a long time at the traffic lights.
Furthermore, the battery also needs to protect the delicate electrical and electronics by absorbing voltage surges and spikes.
BASIC WORKINGS OF A BATTERY
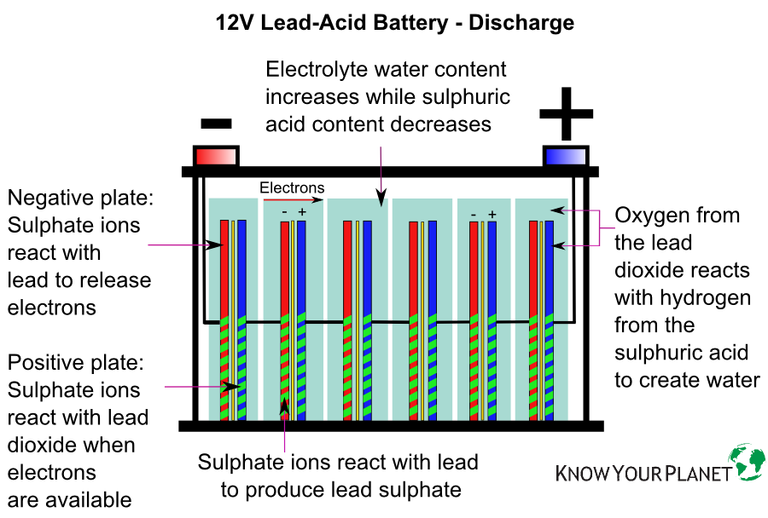
What exactly is a battery? To put it simply, it is an electrochemical device which converts chemical energy to electrical energy.
A basic 12 Volt (V) battery is made up of:
- Six cells that typically produce approximately 2 volts each (depending on type of cells), producing between slightly above 12.0 to 13.2 volts, in total.
- Each cell consists of alternatively charged positive and negative lead (Pb) plates i.e. positive, negative, and so forth. The more plates in a cell, the more current (flow of electricity – Amp) and energy capacity (ampere hours – AH).
- Insulators that are placed between the individual cells.
- The cells are then connected to each other.
- Electrolyte (commonly known as battery acid) which is a sulfuric acid and distilled water solution, is added to the flood the plates.
- The lead plates react chemically to the sulfuric acid, lead sulfate, while the acid is turned into water which in turn is split into hydrogen and oxygen.
- The chemical reaction releases electrons from the negative plates. The electrons (which are negative in charge) flow to the components they need to and return to the battery’s positive terminal and plates. This is what’s known as the electrical current.
- The electrolyte will eventually be diluted through discharge i.e. electrical usage.
- Charging reverses the chemical process.
TYPES OF BATTERIES
1. Wet cell
Also known as the flooded lead-acid cells battery, it was invented way back in 1859. As the name suggests, the cells are inundated in electrolyte and needs to be checked and topped-up as required from time to time, especially in our hot and humid weather.
When stored in a discharged state, the heavier acid molecules sink to the bottom of the battery, causing the electrolyte to stratify (separating into layers of water and acid). When the battery is used again, most of the current will only flow around this area, wearing out the bottom parts of the plates.
Adios soon enough.
Being filled with liquid also requires the battery to be fixed in an upright position to prevent spillage. While being gradually being phased out as OEM fitment for motorcycles, they are still widely available due to their low cost.
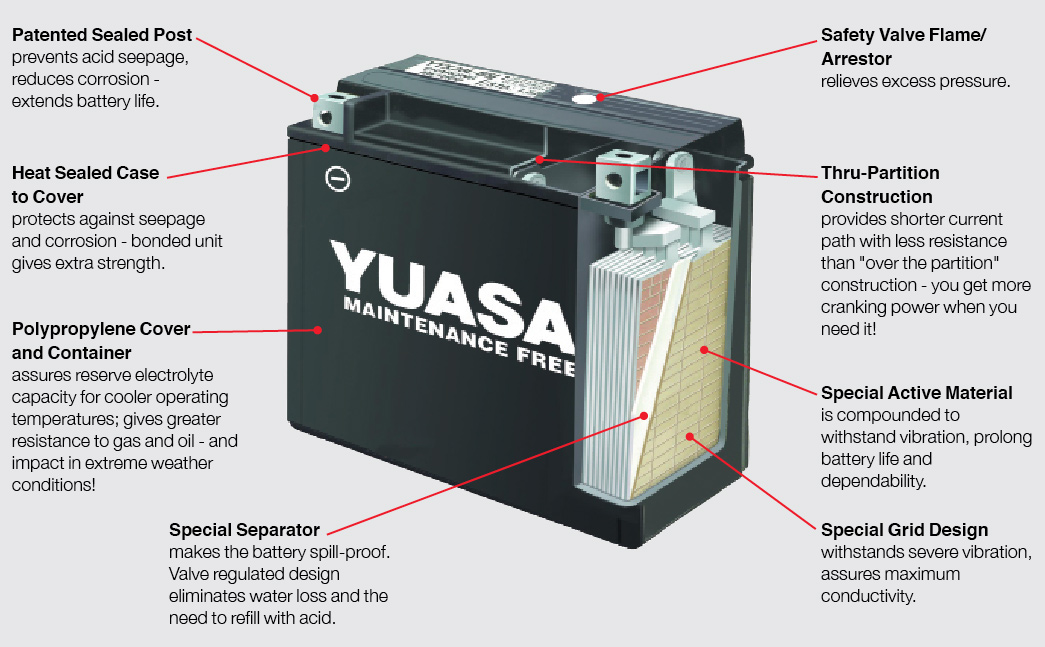
2. Absorbed glass mat (AGM)
The AGM battery is the most common type of OEM fitment nowadays, usually produced by Yuasa for our market.
If you’ve ever replaced your OEM battery to another “maintenance-free” type, especially if it’s from Yuasa or Koyoko, chances are that it’s an AGM.
In an AGM type, the electrolyte is absorbed by a glass fibre mats that sit between cells. The mats greatly reduce evaporation and doesn’t require topping up, consequently. The electrolyte becomes the separator material, allowing the plates to be compressed together, increasing energy density compared to wet cells or gel batteries.
To reduce gas pressure build up in the casing when overcharged or discharged, the AGM battery includes a one-way blow-off valve. Due to that feature, AGM batteries also belong to the group called “valve regulated lead-acid” (VRLA) designs.
Another advantage of the mats is that the electrolyte is held in place and doesn’t slosh around or stratify like in wet cells, which means the battery could be mounted in different positions.
AGM batteries arguably have shorter lifespans since they have higher acid contents to increase standby voltage and lower water loss rate. If your AGM battery shows more than 12.56 V in an open circuit (battery not connected to anything on the bike), it means there’s more acid content. Again, while this is normal for the AGM type, it may not live long.
3. Gel battery
A gel battery, also known as gel cell, is A VRLA battery uses gellified electrolyte. Sulfuric acid is mixed with fumed silica, resulting in an immobile, gel-like mass.
Since there’s no liquid involved, it doesn’t need to be kept upright, electrolyte evaporation is reduced, there’s no spillage and corrosion problems. Besides that, it is more shock and vibration resisitant.
4. Lithium-Ion
Li-Ion batteries are all the rage now, despite not being fitted on a widespread basis due to cost. To describe the functions of a Li-Ion battery requires an entire article by itself!
To surmise, however, the advantages of the Li-Ion compared to VRLAs are mainly substantial weight savings and better cranking pressure (CCA – Cold Cranking Ampere). Cranking pressure denotes how much stored energy is available for cranking up the engine, besides supporting the electronics and electrical systems and accessories.
Disadvantages of Li-Ion?
A discharged Li-Ion battery can’t be brought back to life unlike a VRLA. Additionally, you need to use chargers that are compatible with Li-Ion batteries, although you may also use that charger for VRLA batteries.
That’s it for Part 1, the basics. We’ll cover the subject of maintenance in the next edition.